Ocean Resource Privatization
Articles Posted by Date
The New England groundfish debacle (Part III): who or what is at fault? Nils E. Stolpe/FishNet
NILS STOLPE: The New England groundfish debacle (Part IV): Is cutting back harvest really the answer?
While it’s a fact that’s hardly ever acknowledged, the assumption in fisheries management is that if the population of a stock of fish isn’t at some arbitrary level, it’s because of too much fishing. Hence the term “overfished.” Hence the mandated knee jerk reaction of the fisheries managers to not enough fish; cut back on fishing. What of other factors? They don’t count. It’s all about fishing, because fishing is all that the managers can control; it’s their Maslow’s Hammer. When it comes to the oceans it seems as if it’s about all that the industry connected mega-foundations that support the anti-fishing ENGOs with hundreds of millions of dollars a year in “donations” are interested in controlling. Read the article here
-
Recent Posts
-

Coast Guard searches for missing fisherman off Montauk, N.Y
NEW YORK – The Coast Guard is searching for a missing fisherman who went overboard 16 nautical miles south of Montauk Point, New York, Saturday. Coast Guard Sector Read More » -
Athearn Marine Agency Boat of the Week: 101′ Steel Shrimper, 95k capacity freezer hold, 3508 Cat Diesel
To review specifications, information, and 17 photos’, >click here<, To see all the boats in this series >click here< 12:25 Read More » -

EDF tells NOAA Get multiple buyers for Carlos Rafaels assets, more monitoring
Jim Kendall chuckled as he attempted to grasp the words to describe a letter crafted by Environmental Defense Fund, which it sent to NOAA.,,, The letter Read More » -

Days of chopping off fishing boats over; DFO to increase maximum inshore vessel length to 49’11
“The days of chopping off boats are over. This is a massive victory for inshore harvesters in what is now the under 40’ fleet,” says Jason Read More » -
Closure – Body of sunken El Jefe boat captain recovered
The body of the captain of a fishing boat that sank earlier this week in the Ambrose Channel was recovered Friday afternoon, authorities said. A private Read More » -

When All Hell Breaks Loose: Years after Deepwater Horizon, Offshore Drilling Hazards Persist
This is part one of a three-part investigation into offshore drilling safety. >Read part two here. Read part three here.< They are known as the “last Read More » -

Fishing tech all set to net the catch of the day and nothing else
Dom Talijancich has every available sensor technology on board his 24-meter trawler, FV Mako. Everything but a camera and AI system that identifies what type of Read More » -
Odoriferous : Trident engineers address odor issues at plant
Residents throughout town have been noticing the smell since early July. “My wife was commenting on it just the other day,” said Jerry Pirtle, a local Read More » -
23year Old Lady Born Without Vagina Has One Made Out Of Fish Skin (Photos)
A 23-year-old university student who was born without a vagina, has become the first in the world to undergo pioneering surgery that constructed a new vaginal Read More » -

Crews in Colliers carefully dismantle shipwrecked fishing vessel
A ship that sank in Newfoundland’s Conception Bay 16 years ago is now being demolished and will be recycled, according to the Canadian Coast Guard. The Read More » -
Frank Mirarchi – A Scituate fisherman’s perspective on government’s fisheries management
Thank you for the editorial “White House puts politics ahead of fishery science” published on Tuesday, May 26. You have provided your readers an insight into Read More » -
Few waters contain a more baffling hodgepodge of fishing rules than the Florida Keys
Recreational and commercial fishing in South Florida waters falls under the jurisdiction of three government agencies: The federal South Atlantic Fishery Management Council and the Gulf Read More » -

Cox’s Cove fisherman selling cod to outside markets
Rick Crane was back home Friday after a solo four-day road trip, less than a hour of which was spent selling high-grade catch dockside in Quebec. Read More » -

Seattle-based Alaska crab fleet alerted to new hazard: They’re carrying heavier pots
Alaska crab boats carry stability reports meant to guide the safe loading of up to several hundred crab pots that may be used to bring in Read More » -
Sitka Tribal Government Disappointed With ADF&G
(SitNews) Ketchikan, Alaska – Sitka Tribe of Alaska said they are disappointed in the recent decision by the Alaska Department of Fish and Game to allow Read More » -
Athearn Marine Agency Boat of the Week: 58′ Steel Scalloper/Longline, 550HP Iveco Diesel
Specifications, and information and 19 photos click here To see all the boats in this series, Click here 11:11 Read More » -

Northern Shrimp quota slashed – Who will get the remaining shrimp quota? Who gets to financially survive?
The fights were starting to brew on Friday, as word spread of cuts to northern shrimp quota for 2017. Lean times in the Newfoundland and Labrador Read More » -

Blood, guts and albacore -‘Battlefish’ series gives glimpse into gritty world of West Coast commercial albacore fishing
It’s blood, hooks, aching backs and sun-burnt skin. And cussing. Lots of cussing. A new Netflix series, “Battlefish,” debuted Friday, Sept. 21, giving audiences a raw Read More » -

Are shrimpers abandoning ship?
When Tam Nguyen’s family immigrated to Port Arthur more than 40 years ago, her father made a living owning and operating shrimp boats, the same thing Read More » -
West Coast: Caught in the Sardine Shortage Net
A debate is on about whether the drop is cyclical or a sign of more dire shortages to come. But whatever the reason, commercial fishers, retailers, Read More » -

Harwood grad becomes Alaska salmon boat captain
Jessica Normandeau bought a boat. And not just any boat, a 32-foot Bristol Bay gillnetter, designed for salmon fishing. After graduating from Harwood Union High School Read More » -

‘Our livelihood is taken from us’: Couple feels left out of William’s Harbour resettlement
The Russells have fished out of William’s Harbour for 28 years — from a house that’s been in their family for generations — but they aren’t getting compensation Read More » -
Video – Big boom for tiny shrimp in Petersburg, Alaska
Alaska is home to one of the most prized species of salad shrimp, Pandalus borealis. In Petersburg, Tonka Seafoods has just three boats permitted for salad shrimp fishing, Read More » -
Third Time in Three Years – Pesticides Believed to be Cause of Fish Kills in Canada
For the third time in three years, dead fish have been spotted rising to the surface of Barclay Brook where thousands of fish died in 2011 Read More » -
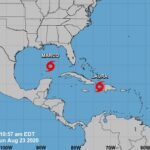
Tropical Storm Media Advisories for Laura and Marco
Marco expected to become a Hurricane today as it enters the Central Gulf Of Mexico, Life-Threatening storm surge and Hurricane Force Winds expected along portions of Read More »
-
Archives
- May 2024 (14)
- April 2024 (191)
- March 2024 (209)
- February 2024 (192)
- January 2024 (211)
- December 2023 (188)
- November 2023 (202)
- October 2023 (180)
- September 2023 (101)
- August 2023 (241)
- July 2023 (237)
- June 2023 (211)
- May 2023 (235)
- April 2023 (210)
- March 2023 (215)
- February 2023 (179)
- January 2023 (187)
- December 2022 (178)
- November 2022 (187)
- October 2022 (190)
- September 2022 (177)
- August 2022 (203)
- July 2022 (186)
- June 2022 (184)
- May 2022 (186)
- April 2022 (190)
- March 2022 (219)
- February 2022 (167)
- January 2022 (192)
- December 2021 (191)
- November 2021 (182)
- October 2021 (196)
- September 2021 (197)
- August 2021 (205)
- July 2021 (221)
- June 2021 (211)
- May 2021 (221)
- April 2021 (204)
- March 2021 (202)
- February 2021 (188)
- January 2021 (195)
- December 2020 (193)
- November 2020 (181)
- October 2020 (204)
- September 2020 (195)
- August 2020 (189)
- July 2020 (205)
- June 2020 (194)
- May 2020 (225)
- April 2020 (218)
- March 2020 (216)
- February 2020 (209)
- January 2020 (233)
- December 2019 (227)
- November 2019 (240)
- October 2019 (241)
- September 2019 (241)
- August 2019 (270)
- July 2019 (288)
- June 2019 (270)
- May 2019 (263)
- April 2019 (223)
- March 2019 (210)
- February 2019 (155)
- January 2019 (117)
- December 2018 (216)
- November 2018 (169)
- October 2018 (218)
- September 2018 (247)
- August 2018 (258)
- July 2018 (259)
- June 2018 (250)
- May 2018 (251)
- April 2018 (247)
- March 2018 (266)
- February 2018 (256)
- January 2018 (278)
- December 2017 (309)
- November 2017 (281)
- October 2017 (288)
- September 2017 (275)
- August 2017 (284)
- July 2017 (287)
- June 2017 (273)
- May 2017 (276)
- April 2017 (275)
- March 2017 (300)
- February 2017 (252)
- January 2017 (288)
- December 2016 (263)
- November 2016 (268)
- October 2016 (287)
- September 2016 (284)
- August 2016 (293)
- July 2016 (286)
- June 2016 (273)
- May 2016 (246)
- April 2016 (267)
- March 2016 (260)
- February 2016 (265)
- January 2016 (269)
- December 2015 (266)
- November 2015 (281)
- October 2015 (289)
- September 2015 (286)
- August 2015 (298)
- July 2015 (294)
- June 2015 (329)
- May 2015 (316)
- April 2015 (317)
- March 2015 (324)
- February 2015 (301)
- January 2015 (332)
- December 2014 (322)
- November 2014 (330)
- October 2014 (382)
- September 2014 (340)
- August 2014 (347)
- July 2014 (376)
- June 2014 (401)
- May 2014 (344)
- April 2014 (341)
- March 2014 (375)
- February 2014 (374)
- January 2014 (360)
- December 2013 (294)
- November 2013 (372)
- October 2013 (391)
- September 2013 (461)
- August 2013 (566)
- July 2013 (526)
- June 2013 (447)
- May 2013 (475)
- April 2013 (508)
- March 2013 (459)
- February 2013 (352)
- January 2013 (265)
- December 2012 (206)
- November 2012 (168)
- October 2012 (187)
- September 2012 (126)
- August 2012 (4)
Tags
Alaska Alaska Department of Fish and Game Athearn Marine Agency Atlantic States Marine Fisheries Commission Boat of the Week BOEM Brexit British Columbia California canada Coast Guard commercial fisherman commercial fishermen commercial fishing Coronavirus Department of Fisheries and Oceans DFO Dungeness crab FFAW FISH-NL Gulf of Maine Gulf of St. Lawrence lobster Louisiana maine Maine Department of Marine Resources massachusetts National Marine Fisheries Service new-england-fishery-management-council Newfoundland and Labrador New Jersey NMFS NOAA North Atlantic right whale North Carolina Fisheries Association North Pacific Fishery Management Council Nova Scotia Obituary offshore wind offshore wind farm offshore wind farms Oregon Scotland United Kingdom weekly update
Comments
- Joel Hovanesian on It’s Been a Brutal Year for Offshore Wind — Despite Analysts’ Best Guesses
- Dennis Haldane on Commercial Fisherman Gainhart (Bud) Samuelson Junior, 77, of Petersburg has passed away
- Garnet Sullivan on Man accused of assaulting conservation officer after elvers bust in downtown Dartmouth
- B on 3 New York wind farms scrapped – Cost implications for Mass., Conn., and R.I.
- Cindy on More things to worry about by Jerry Leeman
- Mark on More things to worry about by Jerry Leeman
- Joel Hovanesian on Where Have All The Right Whales Gone?
- Sid Hounsell on BREAKING: FFAW AND ASP REACH AGREEMENT TO GET SNOW CRAB FISHERY STARTED
- Sid Hounsell on BREAKING: FFAW AND ASP REACH AGREEMENT TO GET SNOW CRAB FISHERY STARTED
- Scott on California’s ocean salmon fishing season closed for second year in a row
- Fran Szymanek on Offshore Wind Electrical Substations; The Secret, Silent Killers by Jim Lovgren
- Nils Stolpe on Time to save the Right Whale from the Green-Left
- Joel Hovanesian on Time to save the Right Whale from the Green-Left
- Chris Iversen on California – Crabbers likely to use new gear next season
- Nils Stolpe on Time to save the Right Whale from the Green-Left
- John Harrison jr on NOAA/NMFS Ignores Dangerous Sound Levels from Pile Driving – By Jim Lovgren
- Chip J on Overspreading Since the Seventies
- borehead - Moderator on Time to save the Right Whale from the Green-Left
- Mike Jacobs on Time to save the Right Whale from the Green-Left
- Joel Hovanesian on East End fishermen uneasy over wind farm South Fork Wind
- Jason taylor on Mi’kmaw fishers say DFO officers left them to walk for hours at night after seizing boots, phones
- Brick Wenzel on East End fishermen uneasy over wind farm South Fork Wind
- Chris Kinder on ENGO Sues UK Government Over International Fishing Quotas
- borehead - Moderator on Mystic Aquarium (the Whale People) expands offshore wind exhibit with youth in mind
- Kath on Mystic Aquarium (the Whale People) expands offshore wind exhibit with youth in mind
- John Harrison jr on Commercial fishermen react to MFC mullet decision
- borehead - Moderator on The CARES Act: Lengthy Process, Little to Show for Connecticut Fisheries
- Randall on The CARES Act: Lengthy Process, Little to Show for Connecticut Fisheries
- Oscar navarrete on Sam Parisi asks, How Accurate is NOAA and NOAA Fishery Survey Science?
- Oscar navarrete on Sam Parisi asks, How Accurate is NOAA and NOAA Fishery Survey Science?
-
Facebook



































Has anybody submitted comments on this issue?
http://www.regulations.gov/#!docketDetail;D=FDA-2011-N-0899
I just submitted these comments. Anybody is welcome to use all or part of them. Just change the first sentance.
I am Chris McCaffity, a commercial fisherman and consumer of seafood. I am deeply concerned about the FDA’s plan to approve Genetically Engineered Salmon for mass production and human consumption. The corporation making these mutated fish claims the unnatural species will never contaminate wild stocks. There is a long history of well-intentioned medaling with Mother Nature having unintended consequences with severe negative impacts when unexpected things like natural disasters occur. Floods could breach holding ponds and carry genetically mutated species into wild waterways. Birds of prey could pick up a mutant fish and drop it in a nearby body of water. Somebody working with these frankenfish could decide to introduce them into the wild to “help” natural fish. There are many ways this kind of experiment could go horribly wrong.
There are much better ways to feed the world a dependable supply of wild-caught seafood and farm-raised species. Here are three examples.
1. Use Artificial Reef habitat to enhance barren seafloor and increase the total bio-mass of marine life an area can support. This would be the perfect union of aquaculture and wild-caught seafood that lives free and self-sufficient until harvested.
2. Collect unused parts of cleaned seafood from fish houses and markets to be processed into feed pellets for farm-raised species. This would help solve the problem of using 3 to 7 pounds of wild seafood to produce one pound of farm-raised product.
3. Properly manage quotas for wild stocks of seafood so that they are fully harvested with very little or no Regulatory Discards that currently waste tons of seafood annually.
I respectfully ask those with the power to approve Genetically Engineered Salmon NOT TO DO SO. Please consider the possible unintended consequences of and positive alternatives to taking this chance with our food supply and native marine life.
Thank you for considering my public comments. [email protected]